Degenerative Mitral Regurgitation
Degenerative MV disease frequently have leaflet prolapse due to elongation or rupture of the chordal apparatus,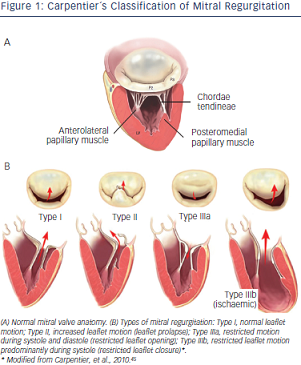 resulting in varying degrees of MV regurgitation due to leaflet malcoaptation during ventricular systole. Physiopathology of systolic flow reversal into the left atrium leads to atrial dilatation/fibrillation, ventricular function impairment and dilatation, secondary pulmonary hypertension and risk of sudden death.
resulting in varying degrees of MV regurgitation due to leaflet malcoaptation during ventricular systole. Physiopathology of systolic flow reversal into the left atrium leads to atrial dilatation/fibrillation, ventricular function impairment and dilatation, secondary pulmonary hypertension and risk of sudden death.
Surgical Treatment
There are several types of degenerative MV regurgitation19,20
(see Figure 1). Current guidelines recommend MV repair when patients develop New York Heart Association (NYHA) class II symptoms, any deterioration in left ventricular function or an end-systolic diameter of 4.5 cm.21 Recent evidence suggests that the best outcomes after repair of severe DMR are achieved in asymptomatic or minimally symptomatic patients, who are selected for surgery soon after diagnosis on the basis of echocardiography.22
Valve repair in patients with degenerative MV disease is associated with an improved quality of life with less morbidity as well as better long-term survival as opposed to replacement. Recent guidelines on valvular heart disease21 contain a reported mortality for isolated MV repair of 1.6–2.1 % and 4.3–7.8 % mortality for MV replacement. More than 80 % of the patients are free from re-operation at five years.
However, even in developed countries, MV replacement remains frequent in this setting. In the Euro Heart Survey, repair rates were documented around 50 %, meaning that MV replacement continues to be performed far too frequently in the modern era of reconstructive valve surgery.23
MV repair for degenerative disease follows two fundamental principles:
Long-term survival following MV repair is similar to age matched controls if the operation is performed before the onset of symptoms, ventricular dysfunction or atrial fibrillation.25
Percutaneous Edge-to-Edge Repair
To date, up to 14,000 patients have been treated with the Mitraclip device worldwide, with nearly 2,000 enrolled in prospective clinical trials. The majority of patients included were considered at high peri-operative risk for MV surgery. After CE mark in 2008, numerous patients have been recruited in several studies. In 2013, the Food and Drug Administration (FDA) approved the Mitraclip device for symptomatic degenerative mitral valve regurgitation for patients at prohibitive risk for MV surgery by a heart team.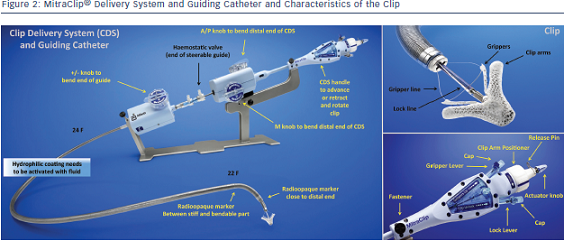
The Mitraclip system consists of a steerable guide catheter and a clip delivery system (CDS), which includes the clip attached at the end of the CDS (see Figure 2).26,27 The guide catheter is 24 Fr proximally, and tapers to 22 Fr at the point where it crosses the interatrial septum (see Figure 2). These steering controls allow the operator to manoeuvre the clip over the MV. The clip is Dacron-covered with two clip arms that are opened and closed by control mechanisms on the CDS. Leaflet tissue is secured between the clip arms and opposing grippers (see Figure 2). The clip arms are then closed to zero degrees, then locked to effect and maintain coaptation of the two leaflets.28 Technical success is achieved in a high number of procedures.29,30 In this technically demanding procedure, the co-operation and communication between the operators and the guidance by transoesophageal echocardiography (see Figure 3) are of great importance. Therefore, the effect of learning curve and co-operation between heart team members could impact on the clinical outcome in terms of MR reduction and complication rates.
The safety and efficacy of the percutaneous edge-to-edge with Mitraclip was initially tested in the Endovascular Valve Edge-to-Edge REpair Study (EVEREST I),15 and then compared with surgery in the randomised standard-risk patients EVEREST II trial.31 Procedural success was defined as successful and stable Mitraclip placement with residual MR ≤2+ on discharge. However, most of the patients enrolled in the EVEREST trials had DMR and 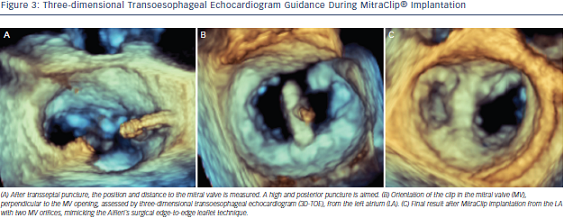 all patients were surgical candidates. The real-world setting is very different from the original EVEREST trials – most of the patients treated with MitraClip are at high surgical risk and MR is more often functional rather than degenerative (see Table 1).
all patients were surgical candidates. The real-world setting is very different from the original EVEREST trials – most of the patients treated with MitraClip are at high surgical risk and MR is more often functional rather than degenerative (see Table 1).
Of note, during the implantation of MitraClip, there is often a compromise between complete reductions of MR and resultant mitral stenosis by placing further clips. Several studies have reported that in these patients, significant improvement in symptoms can still be achieved despite a complete resolution of MR.30,32,33
EVEREST II trial31 included 74 % patients with FMR randomised to surgery or Mitraclip treatment. Latest five-year data have recently been presented at the EuroPCR2014, Paris, France. Mean age in the DMR Mitraclip group was 67 years, 45 % of the patients were in NYHA class III/IV and mean ejection fraction was 64 %. Adverse event rates at 30-days (cardiac, vascular, renal, neurological) were low (25 %) compared with surgical DMR patients (33 %). At one-year, nine patients experienced single leaflet device attachment but no embolisation was observed. From one to five years follow-up, no further single device attachment or embolisation was observed. Survival rates at five years were similar in the Mitraclip and surgical groups (89.4 % versus 85.9 %, respectively). There was a concern on the need of re-operation for the Mitraclip patients. At one-year after the procedure,  75 % of the Mitraclip patients were free from re-operation/MV surgery compared with 100 % of the surgical group. However, from one to five years follow-up, 69 % Mitraclip patients were free from re-operation/MV surgery compared with 96 % of the surgical group, meaning that beyond one-year they observed comparable success rates for surgery and Mitraclip when index Mitraclip procedure was successful (92.2 % versus 95.8 % at five years).
75 % of the Mitraclip patients were free from re-operation/MV surgery compared with 100 % of the surgical group. However, from one to five years follow-up, 69 % Mitraclip patients were free from re-operation/MV surgery compared with 96 % of the surgical group, meaning that beyond one-year they observed comparable success rates for surgery and Mitraclip when index Mitraclip procedure was successful (92.2 % versus 95.8 % at five years).
MR grade at five years follow-up, was ≤2+ in 81 % of the Mitraclip patients and in all surgical patients (100 %), and both groups exerted reduction of left ventricular (LV) end-diastolic diameters. NYHA I/II functional class at five years was present in DMR in 95 % of the Mitraclip patients compared with 97 % of surgical survivors at five years. Mitraclip patients had comparable stability of mitral annular dimensions at five years compared to baseline (4.0 cm versus 3.9 cm, p=0.18). Improved clinical performance in the six-minute walking test have also been reported.34 In the EVEREST II high surgical risk cohort, a 55 % reduction of the annual rate of hospitalisations was observed (0.71–0.32, p=0.006) after Mitraclip implantation.