Low-flow, Low-gradient Aortic Stenosis
Low-flow, Low-gradient Severe Aortic Stenosis with Reduced Left Ventricular Ejection Fraction
Definition
Low-flow, low-gradient severe AS with reduced LVEF, or classical low-flow, low-gradient AS,19 is defined by a MG<40 mmHg or VAS<4 m/s, AVA≤1.0 cm2, and a LVEF<40–50 %.9–11 Inherent in the definition is that flow is reduced, although no specific criteria are included in the guidelines. Individual studies have used a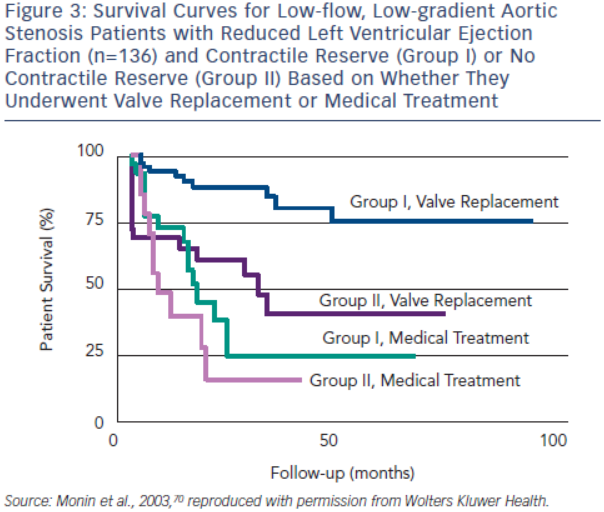 cardiac index <3.0 l/min/m2 or SVi <35 ml/m2,18,19 although measures such as transvalvular flow rate (<200–250 ml/s) may be more appropriate. These patients represent a diagnostic and management challenge since the severity of the underlying valve stenosis is often unclear, AVR is associated with a high operative risk and the benefits of AVR to alleviate symptoms or significantly improve prognosis may be limited in some patients.16–19,54,69–71
cardiac index <3.0 l/min/m2 or SVi <35 ml/m2,18,19 although measures such as transvalvular flow rate (<200–250 ml/s) may be more appropriate. These patients represent a diagnostic and management challenge since the severity of the underlying valve stenosis is often unclear, AVR is associated with a high operative risk and the benefits of AVR to alleviate symptoms or significantly improve prognosis may be limited in some patients.16–19,54,69–71
True Versus Pseudo-severe Aortic Stenosis
AVA, whether measured by continuity equation or orifice planimetry, is affected by flow and becomes smaller when transvalvular flow is reduced.72–74 This phenomenon has been attributed to flow dependence of the flow convergence,75–77 flow-mediated effects on the vena contracta spatial velocity profile,76 reductions in anatomic orifice area due to smaller flow-mediated forces,78,79 or a combination of these factors. The potential effect is that AVA may appear only mild or moderately stenotic when assessed under normal flow conditions, but potentially severe when assessed under low flow conditions. The challenge when encountering the patient with low-flow, low-gradient severe AS with reduced LVEF is to distinguish the patient with ‘true’ severe AS (TSAS), where the valve is severely stenotic and afterload mismatch exists, from the patient with ‘pseudo-severe’ AS (PSAS), where the underlying valve is only mild or moderately stenotic, but appears severe due to low flow and the inability of a poorly functioning ventricle to sufficiently open the valve. AVR is likely beneficial in the TSAS patient, but may not be beneficial in the PSAS patient since the valve is not the primary problem. PSAS is reported to be present in 5–35 % of these patients depending on the specific diagnostic criteria used.69,70,80–82
TSAS and PSAS can be distinguished by augmenting transvalvular flow using dobutamine stress echocardiography (DSE) and re-evaluating the valve haemodynamics at a normal flow state. A commonly used DSE protocol employs an initial infusion of 2.5–5.0 ug/kg/min, followed by incremental infusions of 2.5–5.0 ug/kg/min at 5–8 minute stages to a maximum dose of 20 ug/kg/min.71,82 This low dose, longer stage protocol minimises the potential for myocardial ischaemia and allows for steady state acquisition of LVOT and transvalvular velocities. Patients with TSAS and an underlying ‘fixed’ valve stenosis would be expected to have little change in AVA and a large increase in MG during DSE. In contrast, patients with PSAS would be expected to have an increase in AVA outside of the severe range and only a small increase in MG due to the larger AVA.
Criteria used to diagnose TSAS have included a DSE peak MG>30–40 mmHg, an AVA increase <0.3 cm2, and/or a DSE peak AVA≤1.0–1.2 cm2.18,19,69,70,80–82 PSAS is suggested if the DSE peak AVA is >1.0–1.2 cm2 and the peak MG is ≤30 mmHg or the AVA increase is ≥0.3 cm2. In the AHA/ACC valve guidelines, AVR is considered reasonable in symptomatic patients with low-flow, low-gradient severe AS with reduced LVEF if VAS is ≥4.0 m/s or MG is ≥40 mmHg with an AVA≤1.0 cm2 at any time during DSE (IIa recommendation).9 While this criteria is specific for TSAS, many patients with TSAS have a MG<40 mmHg during DSE because of poor contractile reserve or inherent inconsistencies in the criteria for severe AS.69,82,83 A lower cut-point for MG (i.e. >30 mmHg) may be justified to avoid underdiagnosing TSAS patients in whom the prognosis without AVR is extremely poor.69–71,82,83
A significant limitation to distinguishing TSAS and PSAS patients relates to the marked inter-individual variability to which flow augmentation occurs with DSE.74,82 Some patients have very little increase in transvalvular flow during DSE and a low flow state persists. Other patients have a large increase in transvalvular flow with supranormal flow rates. Since the increase in MG and AVA are largely dependent on the magnitude of flow augmentation, interpretation of the underlying AS severity without consideration of the transvalvular flow is problematic. To address this issue, the projected AVA (AVAproj) at 250 ml/s has been proposed to standardise the calculation of AVA at a normal flow rate and better distinguish TSAS and PSAS.82,83 Transvalvular flow rate and AVA generally display a linear relationship in AS patients with the slope varying between patients.72,74 AVAproj can be calculated by measuring AVA and transvalvular flow rate at a minimum of two different flow rates during DSE using the equation:
AVAproj = AVArest + VC x (250-Qrest)
where AVArest is the resting AVA, Qrest is the resting mean transvalvular flow rate and VC is the ‘valve compliance’ or slope of the valve area – transvalvular flow relationship.82
In an initial 23 patients undergoing surgical inspection, AVAproj≤1.0 cm2 and an AVAproj≤0.55 cm2/m2 correctly classified 83 % and 91 % of patients, respectively.82 In a larger study of 52 patients, a simplified approach was used to calculate VC that only required the measurement of resting and peak DSE transvalvular flow rate and AVA:
VC = (AVApeak - AVArest)/(Qpeak - Qrest)
AVAproj≤1.0 cm2 correctly classified 92 % of patients, and was superior to DSE peak AVA<1.2 cm2 (77 %), peak MG>30 mmHg (73 %) and AVA increase <0.3 cm2 (60 %).83 However, AVAproj needs to be interpreted cautiously if the increase in transvalvular flow rate is <15 % due to inaccuracies in calculating VC.82
Quantitation of AVC by MDCT may provide an additional technique to potentially distinguish TSAS from PSAS, and be especially helpful in those patients in whom the DSE is non-diagnostic or the augmentation of transvalvular flow is minimal. In 18 patients with classical low-flow, low-gradient AS, AVC≥1,651 AU correctly classified 89 % of patients as having either TSAS or PSAS.84 Gender specific thresholds for identifying TSAS are likely warranted based on studies in AS patients with preserved LVEF (~1,250 AU in woman and ~2,050 AU in men), although this has not been studied in classical low-flow, low-gradient AS.41
Prediction of Clinical Outcome
 Contractile or flow reserve, defined as a 20 % increase in SV with DSE, is a strong predictor of peri-operative mortality rate and long-term survival.69,70 AVR is associated with a peri-operative mortality rate as high as 33 % in patients with no contractile reserve, but only 5–8 % in patients with contractile reserve.54,69,70 In the European Multicentre study, three-year survival of patients with contractile reserve undergoing AVR was 79 %, significantly better than the 35 % survival observed in patients with no contractile reserve (see Figure 3).70 Patients with contractile reserve did significantly better with AVR than medical management (three-year survival <25 %). Patients with no contractile reserve have a high operative mortality rate and poor long-term survival; however, their survival is better with AVR than medical therapy when tested using propensity matched analysis.54
Contractile or flow reserve, defined as a 20 % increase in SV with DSE, is a strong predictor of peri-operative mortality rate and long-term survival.69,70 AVR is associated with a peri-operative mortality rate as high as 33 % in patients with no contractile reserve, but only 5–8 % in patients with contractile reserve.54,69,70 In the European Multicentre study, three-year survival of patients with contractile reserve undergoing AVR was 79 %, significantly better than the 35 % survival observed in patients with no contractile reserve (see Figure 3).70 Patients with contractile reserve did significantly better with AVR than medical management (three-year survival <25 %). Patients with no contractile reserve have a high operative mortality rate and poor long-term survival; however, their survival is better with AVR than medical therapy when tested using propensity matched analysis.54
Furthermore, patients with no contractile reserve have similar improvements in LVEF and New York Heart Association (NYHA) functional class following AVR as patients with contractile reserve.85 In essence, absence of contractile reserve identifies patients with a high operative mortality rate and reduced long-term survival who may still benefit from AVR. In the ESC guidelines, AVR should be considered in symptomatic patients with evidence of flow reserve (IIa recommendation) and may be considered in symptomatic patients without flow reserve (IIb recommendation).10
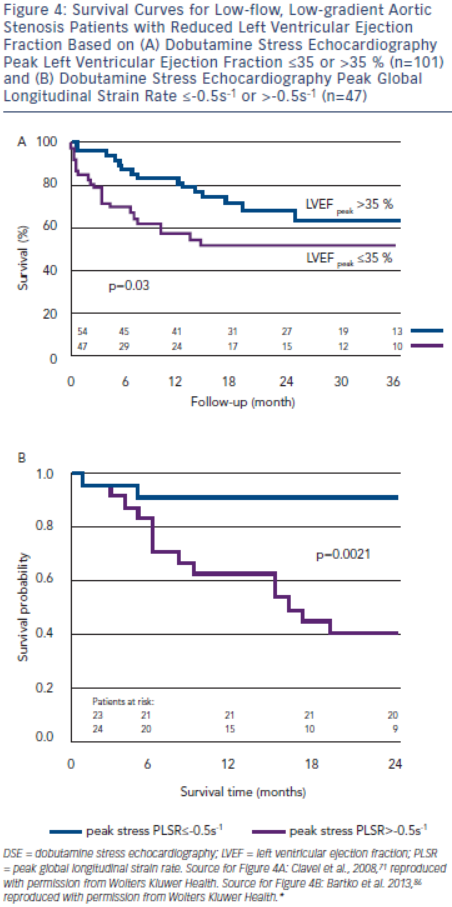
In the True or Pseudo Severe Aortic Stenosis (TOPAS) study of 101 patients with low-flow, low-gradient severe AS with reduced LVEF, independent echocardiographic predictors of reduced survival were AVAproj≤1.2 cm2 and DSE peak LVEF≤35 % (see Figure 4A).71,83 Contractile reserve was not an independent predictor of survival.71 The conflicting findings regarding the predictive value of contractile reserve in TOPAS and the European Multicentre
Study may have related to different DSE protocols or differences in the study population (TOPAS had a greater prevalence of concomitant diseases such as coronary artery disease).70,71 Additionally, DSE peak LVEF is essentially a composite measure of both resting LVEF and ‘contractile reserve’, and may explain the better predictive value of this parameter.
Longitudinal strain parameters are severely impaired in low-flow, low-gradient severe AS with reduced LVEF, but may improve with DSE in a substantial proportion of patients. In a substudy of TOPAS, patients with a DSE peak global longitudinal strain rate (PLSR) <-0.5 s-1 had a two-year survival of 91 %, but only 40 % if DSE PLSR was ≥-0.5 s–1 (see Figure 4B).86 DSE PLSR was the strongest predictor of overall survival and added incremental prognostic information to DSE peak LVEF.
Low-flow, Low-gradient Severe Aortic Stenosis with Preserved Left Ventricular Ejection Fraction
Definition
The criteria for low-flow, low-gradient severe AS with preserved LVEF include a VAS<4 m/s or MG<40 mmHg, AVA≤1.0 cm2, LVEF>50 % and SVi<35 ml/m2.9–11,19
True Versus Pseudo-severe Aortic Stenosis
Patients with low-flow, low-gradient severe AS with preserved LVEF have similar transvalvular flow rates as classical low-flow, low-gradient severe AS.83,87 Since 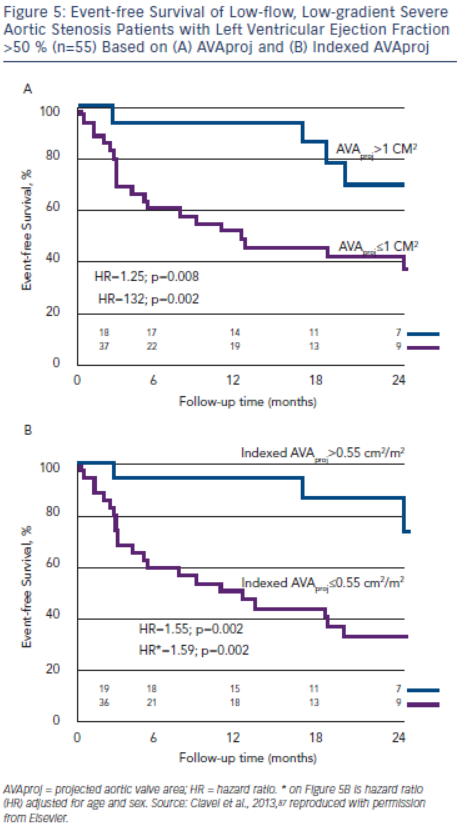 transvalvular flow is reduced, the small AVA may reflect underlying TSAS or PSAS. The prevalence of PSAS is reported to be ≈30 % in these patients, similar to what is observed in classical low-flow, low-gradient severe AS.87 DSE and exercise echocardiography have been used to distinguish TSAS and PSAS in one small study.87 AVAproj≤1.0 cm2 was 94 % accurate in distinguishing the two conditions when compared to surgical inspection, and performed better than DSE peak MG≥30 mmHg (75 %) or ≥40 mmHg (81 %) and DSE peak AVA≤1.0 cm2 (69 %) or ≤1.2 cm2 (63 %). In the AHA/ACC and ESC valve guidelines, AVR is reasonable in symptomatic patients if the clinical, haemodynamic and anatomic data support valve obstruction as the most likely cause of the symptoms (IIa recommendation).9,10 Reserving surgical intervention only for symptomatic TSAS patients appears theoretically sound, although this management strategy has not been tested.
transvalvular flow is reduced, the small AVA may reflect underlying TSAS or PSAS. The prevalence of PSAS is reported to be ≈30 % in these patients, similar to what is observed in classical low-flow, low-gradient severe AS.87 DSE and exercise echocardiography have been used to distinguish TSAS and PSAS in one small study.87 AVAproj≤1.0 cm2 was 94 % accurate in distinguishing the two conditions when compared to surgical inspection, and performed better than DSE peak MG≥30 mmHg (75 %) or ≥40 mmHg (81 %) and DSE peak AVA≤1.0 cm2 (69 %) or ≤1.2 cm2 (63 %). In the AHA/ACC and ESC valve guidelines, AVR is reasonable in symptomatic patients if the clinical, haemodynamic and anatomic data support valve obstruction as the most likely cause of the symptoms (IIa recommendation).9,10 Reserving surgical intervention only for symptomatic TSAS patients appears theoretically sound, although this management strategy has not been tested.
Measurements of AVC using MDCT may have a role in identifying TSAS and PSAS in patients with low-flow, low-gradient severe AS with preserved LVEF. In 451 patients with at least moderate AS, preserved LVEF, normal SVi and concordant MG and AVA measurements, an AVC threshold of 1,274 AU in women and 2,065 AU in men correctly classified 87 % of patients as having moderate AS or severe AS.41 A similar 87 % accuracy was obtained using an AVC indexed to aortic annulus area threshold of 292 AU/cm2 for women and 476 AU/cm2 for men. In a further 172 patients with preserved LVEF and discordant AS measurements (MG<40 mmHg and AVA≤0.6 cm2/m2), ~50 % of patients had an AVC score consistent with moderate rather than severe AS.41 Low flow (SVi <35 ml/m2) was present in ~16 % of these patients; however, results for this subgroup were not provided and DSE results were not reported. Further studies are warranted to determine whether AVC thresholds can be used to distinguish TSAS and PSAS in low-flow, low-gradient severe AS with preserved LVEF.
Prediction of Clinical Outcome
AVAproj and indexed AVAproj are strong predictors of adverse outcome in patients with low-flow, low-gradient severe AS with preserved LVEF (hazard ratio: 1.32 and 1.59, respectively, for each 0.1 cm2 decrease in AVAproj and indexed AVAproj) and is superior to traditional parameters of stenosis severity for predicting outcome.87 More than 90 % of patients with AVAproj>1.0 cm2 or indexed AVAproj>0.55 cm2/m2 remain free of death or AVR at 12 months, whereas only 50 % of patients with AVAproj≤1.0 cm2 or AVAproj≤0.55 cm2/m2 will be free of death or AVR (see Figure 5).87