The Need to Overcome the Morphologic Limitations of Coronary Angiography
All of these morphologic limitations intrinsic to CA inspired the development of realtime high-resolution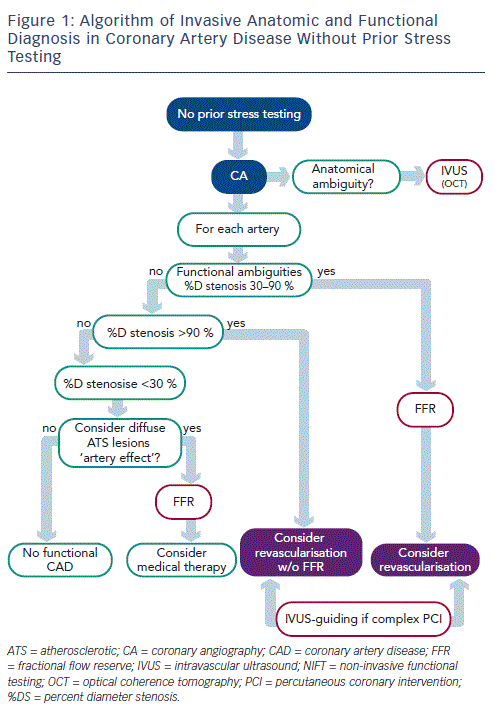 invasive complementary imaging techniques. These cross-sectional images provide full information from the lumen to the complete arterial wall. They enable precise quantification, with the possibility of three-dimensional (3D) reconstruction. Thus in the early 1990s, intravascular ultrasound (IVUS) began to complement the morphologic failings of CA by supplying cross-sectional imagery. Progressive advances have notably concerned improved spatial resolution by increasing the ultrasound frequency (90 micrometer [μm] at 40 megahertz [MHz]). Quantitative analysis thus gained in precision, overall assessment of the arterial wall and its impact on the lumen became clearly defined, and pathological mechanisms at work in the wall can now be rapidly detected and correctly described.22 Analysis of plaque composition is more limited, being based on simple acoustic reflectivity, providing only approximate assessment of the ternary aspect of the usual plaque components (anechogenic or hypoechogenic structures), which merely indicate the absence of a reflective ingredient such as calcium, collagen or elastin.23 IVUS resolves certain endo- or extra-luminal ambiguities with a more or less complex additional image; and precisely detects plaque rupture following acute coronary syndrome, parietal thrombus, intra-plaque or intra-adventitia haematoma and certain ambiguities in the distribution or protrusion of calcification. Alongside the development of complex coronary angioplasty, IVUS has become a tool for selecting, guiding and assessing percutaneous coronary intervention (PCI) with stent implantation.24 Long-term clinical outcome after IVUS-guided stent implantation and long-term mortality in stenting for unprotected left main coronary artery stenosis have improved significantly.25 Taken together, these advantages, completing classical CA diagnosis with precision, have been drawn up into a level IIa/IIb set of recommendations.26
invasive complementary imaging techniques. These cross-sectional images provide full information from the lumen to the complete arterial wall. They enable precise quantification, with the possibility of three-dimensional (3D) reconstruction. Thus in the early 1990s, intravascular ultrasound (IVUS) began to complement the morphologic failings of CA by supplying cross-sectional imagery. Progressive advances have notably concerned improved spatial resolution by increasing the ultrasound frequency (90 micrometer [μm] at 40 megahertz [MHz]). Quantitative analysis thus gained in precision, overall assessment of the arterial wall and its impact on the lumen became clearly defined, and pathological mechanisms at work in the wall can now be rapidly detected and correctly described.22 Analysis of plaque composition is more limited, being based on simple acoustic reflectivity, providing only approximate assessment of the ternary aspect of the usual plaque components (anechogenic or hypoechogenic structures), which merely indicate the absence of a reflective ingredient such as calcium, collagen or elastin.23 IVUS resolves certain endo- or extra-luminal ambiguities with a more or less complex additional image; and precisely detects plaque rupture following acute coronary syndrome, parietal thrombus, intra-plaque or intra-adventitia haematoma and certain ambiguities in the distribution or protrusion of calcification. Alongside the development of complex coronary angioplasty, IVUS has become a tool for selecting, guiding and assessing percutaneous coronary intervention (PCI) with stent implantation.24 Long-term clinical outcome after IVUS-guided stent implantation and long-term mortality in stenting for unprotected left main coronary artery stenosis have improved significantly.25 Taken together, these advantages, completing classical CA diagnosis with precision, have been drawn up into a level IIa/IIb set of recommendations.26
Endocoronary imaging has recently seen the advent of OCT, based on a different physical principle from IVUS and providing much-improved spatial resolution of around 13 μm.27 Unfortunately, the physics underlying this technique means that the wavelengths used cannot penetrate deep in a plaque (not more than 0.5–1.0 millimetres [mm], depending on the type of tissue) thus the advantage of greatly enhanced resolution applies only to the juxtaluminal wall and endoluminal evolutive processes. Analysing the surface of a stented lumen, for example, corresponds exactly to the field of application of OCT. However, its drastic limitation in deep exploration means that it is not an alternative in those applications for which IVUS is especially useful and recommended, such as guiding complex or left coronary angioplasty.28
The Need to Overcome the Functional Limitations of Coronary Angiography
Determining FFR by pressure guide was a critical advance, fully confirmed first experimentally and then clinically over the last 15 years by the work of Pijls and de Bruyne.15,16,29,30 After numerous precise cumulative validations, it has been shown to be an exact technique, specific to all obstructive epicardial lesions. FFR is independent of heart rate, arterial pressure and myocardial contractility. FFR has an unequivocal value of 1.0 in any patient with a normal coronary artery. An FFR 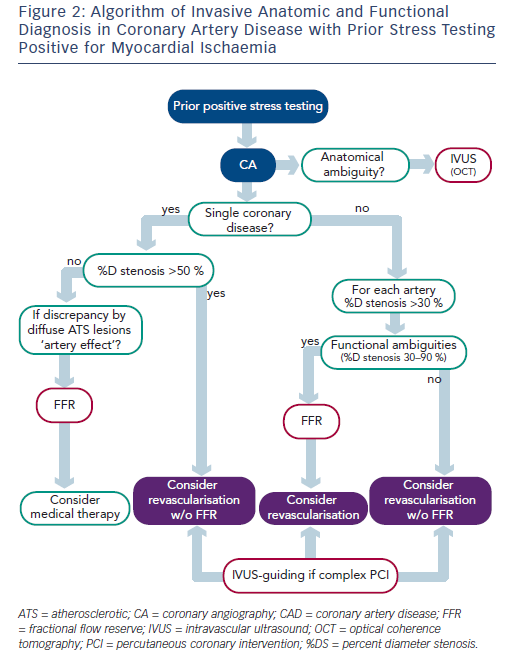 equal to or less than 0.8 precisely determines the functional impact of a significant as compared to a non-significant stenosis with a high degree of accuracy. FFR also takes into account the contribution of any collateral flow to myocardial perfusion, and can be applied in single or multivessel disease, as it does not require having a normal coronary artery to serve as control. It is moreover easy to perform, highly reproductible and can be implemented without difficulty in both diagnostic and therapeutic procedures. The key point in applying this technique is that it is specific to the functional impact of an epicardial stenosis on coronary circulation, independently of the patient’s individual microvascular resistance patterns. In this respect, it is completely different from CFR, which characterises the functional association of the epicardial network with the microvascular resistive network, so that it is not specific to epicardial stenosis. Several studies demonstrated that coronary stenoses of 30–70 % DS are functionally ambiguous, as they may or may not be associated with any myocardial ischaemia.31 The Fractional Flow Reserve Versus Angiography for Multivessel Evaluation (FAME) study of 1,414 lesions assessed by FFR clearly showed that 65 % of tight (50–70 % DS) stenoses and 20 % of 71–90 % DS stenoses were not inducing any myocardial ischaemia (FFR >0.80).32 Only 235 out of 509 patients with multivessel disease as shown on CA (46 %) had functional multivessel disease (≥2 coronary arteries with FFR >0.80). In other words, when an intermediate coronary stenosis (between 30 and 90 %) is detected, its functional implications remain entirely ambiguous. Pressure guide determination of FFR precisely assesses impact on the epicardial network, independently of haemodynamics and particularly of the resistive effect of myocardial microcirculation. Thus FFR is a major stage in determining indications for revascularisation where stenosis shows a functional impact.
equal to or less than 0.8 precisely determines the functional impact of a significant as compared to a non-significant stenosis with a high degree of accuracy. FFR also takes into account the contribution of any collateral flow to myocardial perfusion, and can be applied in single or multivessel disease, as it does not require having a normal coronary artery to serve as control. It is moreover easy to perform, highly reproductible and can be implemented without difficulty in both diagnostic and therapeutic procedures. The key point in applying this technique is that it is specific to the functional impact of an epicardial stenosis on coronary circulation, independently of the patient’s individual microvascular resistance patterns. In this respect, it is completely different from CFR, which characterises the functional association of the epicardial network with the microvascular resistive network, so that it is not specific to epicardial stenosis. Several studies demonstrated that coronary stenoses of 30–70 % DS are functionally ambiguous, as they may or may not be associated with any myocardial ischaemia.31 The Fractional Flow Reserve Versus Angiography for Multivessel Evaluation (FAME) study of 1,414 lesions assessed by FFR clearly showed that 65 % of tight (50–70 % DS) stenoses and 20 % of 71–90 % DS stenoses were not inducing any myocardial ischaemia (FFR >0.80).32 Only 235 out of 509 patients with multivessel disease as shown on CA (46 %) had functional multivessel disease (≥2 coronary arteries with FFR >0.80). In other words, when an intermediate coronary stenosis (between 30 and 90 %) is detected, its functional implications remain entirely ambiguous. Pressure guide determination of FFR precisely assesses impact on the epicardial network, independently of haemodynamics and particularly of the resistive effect of myocardial microcirculation. Thus FFR is a major stage in determining indications for revascularisation where stenosis shows a functional impact.
In 1999 Takagi et al. again attempted to correlate stenosis anatomy (IVUS minimum lumen area [MLA], square millimetre [mm2]) and functional impact (FFR), and reported a 3 mm2 IVUS MLA cut-off as detecting stenosis with FFR <0.75 with 83 % sensitivity and 92 % specificity.332 IVUS MLA value became a functional surrogate index. However, in a recent rigorous study of 267 lesions, Koo et al.34 found no lumen area cut-off reliably associated with FFR <0.80, in agreement with Kang et al.35 In fact, each coronary artery bifurcation displays a fractal geometry, with diameters progressively narrowing in line with the law of conservation of mass35 – thus, no one functional coronary lumen area criterion is conceivable, and no quantitative CA stenosis index can be precisely correlated with and predictive of myocardial functional impact. Another functional scenario to be detected is when an epicardial artery is diffusely infiltrated by atherosclerosis, with diameters that are reduced overall, not letting focal stenoses appear – should any exist, they will be poorly quantified as the reference segment will be seriously underestimated. FFR determined with long pressure-guide pullback provides perfect analysis of this anatomic–functional situation.37Accumulated evidence shows that the functional significance of the coronary stenosis determines the potential benefit of revascularisation in patients with stable coronary artery disease (CAD).38 When an artery in stable CAD is revascularised by FFR-guided CA, the rate of coronary events diminishes.38,39 Moreover, there is serious evidence that medical treatment can be preferable to PCI in patients with stable coronary artery disease.40 The results of the Fractional Flow Reserve-Guided Percutaneous Coronary Intervention Plus Optimal Medical Treatment Versus Optimal Medical Treatment Alone in Patients With Stable Coronary Artery Disease (FAME II) study refine this body of evidence, showing that in stable angina with functionally significant coronary stenosis, FFR-guided PCI associated to optimal medical treatment (evidence-based medicine) reduces the rate of emergency revascularisation compared with optimal medical treatment alone.41