Putting it all together: Aortic Paravalvular Leak
Retrograde transfemoral approach is the optimal strategy for aortic PVL. TEE is used for imaging, with description of the leak on the clock face as described above. Once the leak is crossed with a hydrophilic wire, it is crossed again with a 5 Fr diagnostic catheter (often JR4, MP or Amplatz-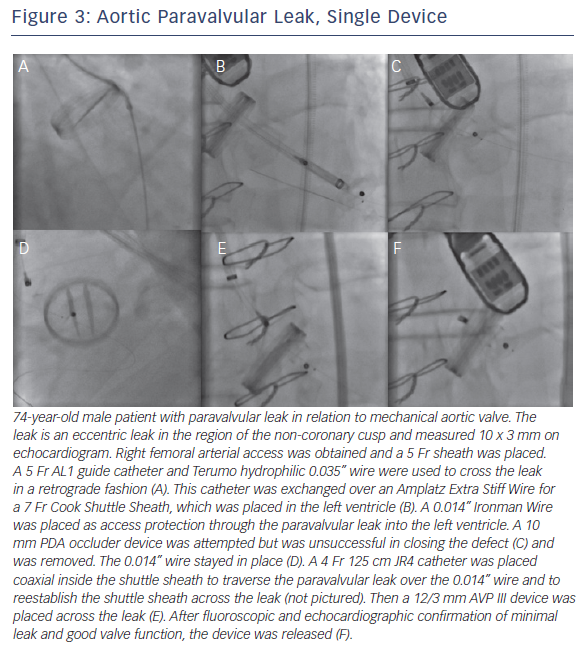
If there is significant tortuosity or difficulty crossing the leak, more support can be obtained by building a rail. A transseptal rail invo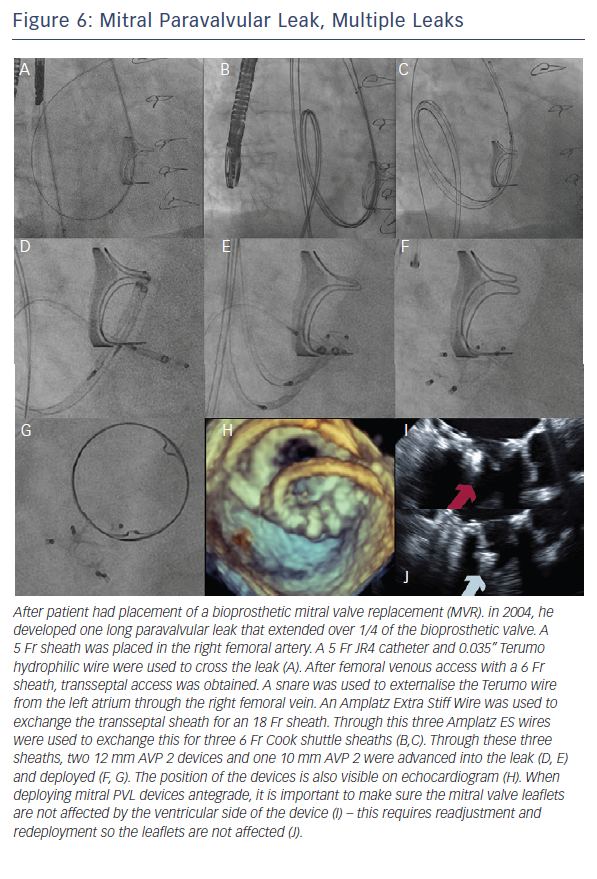 lves transseptal access with subsequent snaring of the original wire within the left atrium. An apical ventricular rail involves left ventricular puncture and having the wire through the apex. Figures 3 and 4 demonstrate aortic PVL closure.
lves transseptal access with subsequent snaring of the original wire within the left atrium. An apical ventricular rail involves left ventricular puncture and having the wire through the apex. Figures 3 and 4 demonstrate aortic PVL closure.
Subsequent aortic angiography is often necessary to rule out coronary compression and evaluate the valve for regurgitation. The valve should not have an increased gradient and should have free-moving leaflets. If the device was implanted in the area of the non-coronary cusp, special attention should be given to the anterior mitral valve leaflet.
Putting it all together: Mitral Paravalvular Leak
After evaluating the leak with TEE, the first approach is to try the simplest approach: retrograde transfemoral approach with a 5 Fr IM or JR4 catheter and a hydrophilic 0.035” guidewire. 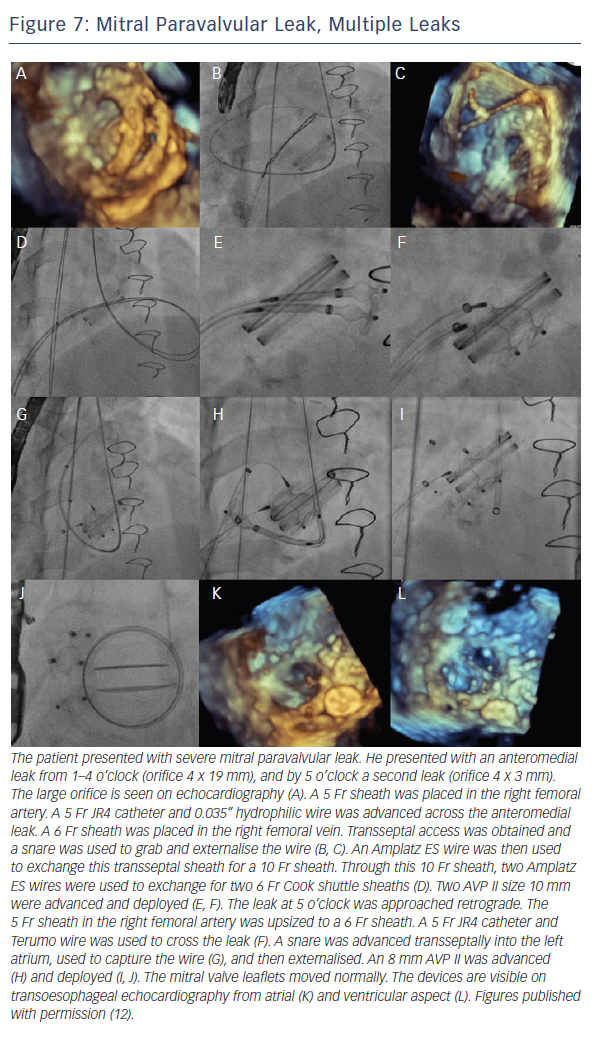 Once this is across the leak, the wire can be exchanged for a stiff wire and then the delivery system (guide catheter or long sheath). The wire is removed, the device is advanced, and then the device is deployed. By deploying the first disc within the left atrium, it is simpler to visualise on TEE. If there is an issue with support, transseptal access with snare can be used to make a rail. As mentioned above, it is important to protect the aortomitral continuity with a catheter whenever possible.
Once this is across the leak, the wire can be exchanged for a stiff wire and then the delivery system (guide catheter or long sheath). The wire is removed, the device is advanced, and then the device is deployed. By deploying the first disc within the left atrium, it is simpler to visualise on TEE. If there is an issue with support, transseptal access with snare can be used to make a rail. As mentioned above, it is important to protect the aortomitral continuity with a catheter whenever possible.
When the retrograde transfemoral approach is not successful, an antegrade transseptal approach can have some benefits. It is important to cross posteriorly to avoid the aorta and superiorly to have enough catheter room to reach both medial and lateral leaks. A similar approach with a 0.035” hydrophilic wire, 5Fr JR4 or MP catheter, stiff wire exchange, and then exchange for delivery system (guide catheter or long sheath) is used. If support is still an issue, a transarterial rail is recommended. Another option is to advance the transseptal sheath through the defect. Some centers have used an Agilis system (St. Jude Corporation) if the catheter is unable to reach the defect or if the puncture site was suboptimal. The advantage that this offers is increased steerability; the disadvantage is a larger transseptal puncture and increased cost of the procedure. Figures 5, 6 and 7 show examples of mitral PVLs.
After the device is placed, TEE should show mobile mitral leaflets, open pulmonary veins, and if the leak was anterior, an unobstructed mitral valve. Angiography is insufficient to make this determination. The authors do no recommend release of a device until these requirements are satisfied.
Device success
There are many ways to characterise device success, but the device should treat the problem it is there to solve – there should be a sizeable decrease in regurgitation and improvement in symptoms. The patient should have a TTE for aortic leak and TEE for mitral leak at 6 months (or earlier if symptoms are present). If there is hemolysis, the hemoglobin/hematocrit should also be monitored.