Methods to Enhance Platelet Inhibition
Studies to overcome the potential delay in platelet inhibition, associated with immediate pre-procedural loading of oral antiplatelet therapy, have been undertaken. The ATLANTIC investigators addressed this question by randomising 1,862 patients presenting within 6 hours of STEMI onset to pre-hospital versus in-hospital treatment with ticagrelor.34 Pre-hospital ticagrelor did not improve pre-PCI coronary perfusion but appeared to be safe and was associated with a reduction in post-procedural AST. The median time between the two loading doses (pre-hospital versus in-hospital) was 31 minutes.
Alternatives to upstream administration of an oral antiplatelet therapy include manipulation of the pharmacokinetic properties of oral agents or use of an intravenous platelet inhibitor. The Mashed Or Just Integral Tablets of ticagrelOr (MOJITO) study35 tested the effect of crushing ticagrelor to accelerate drug absorption and demonstrated a significant enhancement of platelet inhibition 1 hour following drug ingestion. A larger scale trial with clinical endpoints would be necessary to validate these results. Cangrelor is an intravenous adenosine triphosphate (ATP) analogue, which reversibly inhibits the P2Y12 receptor without requiring hepatic conversion.36 The attraction of cangrelor is therefore its very rapid onset of action and short halflife (3–5 minutes) allowing rapid platelet inhibition and quick reversal. Two early trials (Cangrelor versus standard therapy to achieve optimal management of platelet inhibition [CHAMPION]-PCI37 and CHAMPIONPLATFORM38) evaluating cangrelor in patients undergoing PCI failed to show clinical superiority over clopidogrel. The more recent CHAMPION-PHOENIX39 trial randomised 11,145 patients undergoing urgent or elective PCI to intravenous cangrelor or clopidogrel 600 or 300 mg loading (56 % stable angina/18 % STEMI). At 48 hours the rate of composite primary efficacy endpoint (death, MI, ischaemiadriven revascularisation or stent thrombosis) occurred less in the cangrelor group (4.7 % versus 5.9 %) and the rate of stent thrombosis was significantly lower (0.8 % cangrelor versus 1.4 % clopidogrel). Although the study demonstrated benefit with use of cangrelor, there were significant limitations in the trial design, favouring the study drug arm. The control group only received clopidogrel once the anatomy was delineated, and 30 % of the cohort were administered the drug post-PCI. Consequently, the higher rate of peri-procedural MI in the control group is not surprising.
Antiplatelet Strategies in Patients on Oral Anticoagulation
A significant proportion of patients undergoing PPCI may already be anticoagulated on a vitamin K antagonist (VKA) 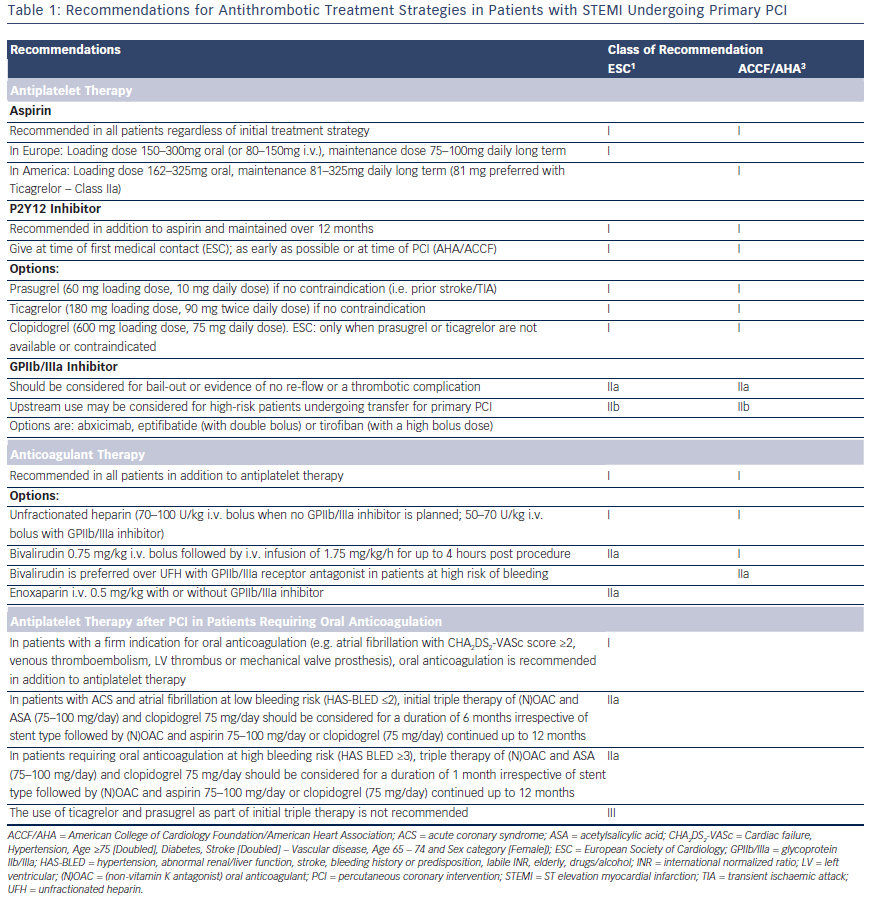 or a non-vitamin K antagonist oral anticoagulants ((N)OAC) at the time of the procedure. These patients are at increased risk of bleeding and often the international normalized ratio (INR) levels are not available. There is no clear evidence on the optimal antithrombotic pharmacotherapy for these patients. The 2014 ESC/EACTS guidelines on myocardial revascularisation1 recommends that PPCI in this setting should be performed via a radial approach with use of additional parenteral anticoagulation regardless of the timing of the last dose of oral anticoagulant. Bivalirudin may be preferred due to its short half-life and should be discontinued immediately after PPCI. GPIs should generally be avoided unless for bail-out situations.
or a non-vitamin K antagonist oral anticoagulants ((N)OAC) at the time of the procedure. These patients are at increased risk of bleeding and often the international normalized ratio (INR) levels are not available. There is no clear evidence on the optimal antithrombotic pharmacotherapy for these patients. The 2014 ESC/EACTS guidelines on myocardial revascularisation1 recommends that PPCI in this setting should be performed via a radial approach with use of additional parenteral anticoagulation regardless of the timing of the last dose of oral anticoagulant. Bivalirudin may be preferred due to its short half-life and should be discontinued immediately after PPCI. GPIs should generally be avoided unless for bail-out situations.
Duration of Dual Antiplatelet Therapy post-PPCI DAPT (aspirin + a P2Y12 inhibitor) is recommended for 1 year in patients undergoing PPCI for STEMI. This recommendation is based on the early CURE study40 (clopidogrel) and is supported by more recent results from TRITON-TIMI 38 (Prasugrel)15 and PLATO (Ticagrelor).17 Regardless of stent type, extended DAPT for 1 year reduces the risk of stent thrombosis, re-infarction and cardiovascular mortality6 with the more potent DAPTs associated with the greatest post-ACS clinical benefit.41 Recent data have highlighted that extended DAPT confers further protection against ischaemic events but at the expense of additional bleeding risk.42 In stark contrast, the Global-Leaders trial (NCT01813435) is currently enrolling patients to either 1 month DAPT with aspirin and ticagrelor, and then ticagrelor monotherapy for 23 month or 12 months DAPT with aspirin and ticagrelor/clopidogrel with aspirin monotherapy between 12 and 24 months. We await the results with interest.
Antiplatelet Therapy after PCI in Patients Requiring Oral Anticoagulation
A proportion of patients on DAPT post-PPCI will have a firm indication for long-term anticoagulation (atrial fibrillation with Cardiac failure, Hypertension, Age ≥75 [Doubled], Diabetes, Stroke [Doubled] – Vascular disease, Age 65 – 74 and Sex category [Female]) [CHA2DS2- VASc] score ≥2, venous thromboembolism, left ventricular [LV] thrombus, mechanical valve prosthesis). Triple therapy with an oral anticoagulant, aspirin 75–100 mg and clopidogrel 75 mg should be limited in duration depending on the clinical setting, thromboembolic risk (CHA2DS2-VASc score) and bleeding risk (HAS-BLED score). The WOEST trial43, which randomised 573 patients either to dual therapy or triple therapy, showed that in patients on oral anticoagulants the use of clopidogrel without aspirin was safe. TIMI bleeding and allcause mortality was lower in the dual therapy group with no increase in the rate of thrombotic events. The 2014 ESC/EACTS guidelines on myocardial revascularisation1 recommends 1 month of triple therapy for ACS patients at high bleeding risk (HAS-BLED >3) and 6 months for patients at lower bleeding risk, followed by dual therapy (oral anticoagulant and clopidogrel or aspirin) for a minimum of 12 months (see Table 1). The use of prasugrel and ticagrelor as part of triple therapy should be avoided44 and gastric protection with a proton pump inhibitor should be implemented.