Myocardial Protection
Some mechanical assist devices protect the myocardium by increasing oxygen delivery and reducing the oxygen demand, thereby preventing myocardial damage. This is achieved by a combination of increasing the aortic pressure and unloading the ventricle. Depending on the device, unloading is achieved through direct unloading of the left ventricle or by decreasing the preload of the left ventricle. Unloading the left ventricle results in a decreased LV end-diastolic pressure and peak LV wall stress, which in turn leads to decreased microvascular resistance, myocardial work load and myocardial oxygen consumption. By decreasing microvascular resistance, coronary blood flow is increased, as the latter is the result of the pressure difference between the proximal and distal vascular bed. In conclusion, the ideal cardiac assist device protects the myocardium by increasing coronary perfusion and decreasing myocardial workload.
Mechanical Assist Devices
Over the past decades, many LV support devices have been developed, such as surgical bridge-to-transplant or destination therapy devices as well as temporary (percutaneous) bridge-to-recovery devices.
During an acute critical presentation, a less-invasive percutaneous approach is preferable as it provides a quick and easy deployment. The ideal device is instantly accessible and should be associated with a low complication rate, as sometimes complications outweigh the potential beneficial effects. Complications associated with any (percutaneous) LV assist device include limb ischaemia, embolisation of atherosclerotic or thrombotic material, stroke, infection and haemolysis. In the following sections, the most common and currently used percutaneous devices are described: the intra-aortic balloon pump (IABP), TandemHeart (Cardiac Assist Inc., Pittsburgh, PA, US), ECMO (extracorporeal membrane oxygenation) and Impella (Abiomed Europe GmbH, Aachen, Germany). These devices differ in insertion technique as well as working mechanism.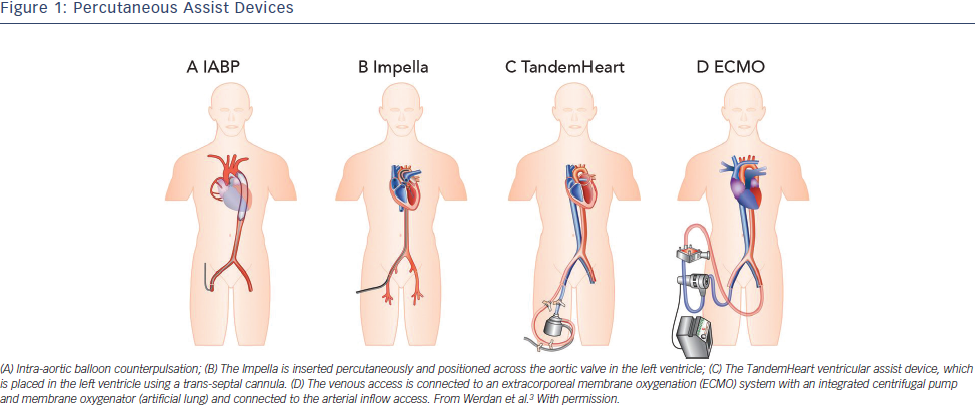
Intra-aortic Balloon Pump
The IABP was, since its introduction in 1968, the most frequently used and broadly available cardiac assist device. The IABP is inserted percutaneously in the femoral artery and the balloon is positioned in the descending thoracic aorta distal to the left subclavian artery and proximal to the renal artery branches (see Figure 1A).3 The balloon is synchronised to the cardiac cycle and is rapidly inflated during diastole and rapidly deflated during early systole by input and removal of helium gas. The IABP is assumed to increase coronary and systemic blood flow due to augmentation of the diastolic blood pressure during inflation of the balloon. Deflation of the balloon decreases myocardial oxygen demand by decreasing afterload and ventricular wall tension, while slightly increasing coronary bloodflow.4 The IABP generates an increase of CO up to approximately 0.3–0.5 l/min. There are several limitations to the IABP. The augmentation of CO is likely to be insufficient for patients with severe CS. Also, to provide haemodynamic support it requires a certain level of LV function. Finally, the function of the IABP relies on synchronisation with the cardiac cycle, which might not be reliable in case of cardiac arrhythmias in critically ill patients.
Impella
The Impella is a micro-axial rotary pump that is placed across the aortic valve expelling aspirated blood from the left ventricle into the ascending aorta (see Figure 1B). Currently, there are three versions of the Impella system available. The Impella 2.5 and Impella CP can provide up to 2.5 l/min and 3.5–4.0 l/min, respectively, and are percutaneously inserted. The Impella 5.0 can deliver up to 5.0 l/min but requires a surgical cutdown of the femoral or axillary artery. The device has a pigtail-catheter at the tip to ensure stable positioning in the left ventricle and to avoid adhering to the myocardium. The direct unloading of the left ventricle is an important feature of the Impella. The unloading effect of the Impella 2.5 was demonstrated by a reduction in end-diastolic wall stress and immediate decrease in pulmonary capillary wedge pressure (PCWP).5–7 The Impella-induced increase in coronary bloodflow probably results from both an increased perfusion pressure and a decreased LV volume-related intra-myocardial resistance. In an experimental setting, haemodynamic support and unloading with the Impella has been demonstrated to reduce infarct size.8 The Impella 5.0 should result in even more unloading due to the substantially larger contribution to overall circulation. In contrast to the IABP, the Impella works independently of LV function and cardiac rhythm.
TandemHeart
TandemHeart is a trans-septal ventricular assist device that is inserted through the femoral vein and right atrium into the left atrium via an atrial septum puncture (see Figure 1C). The outflow cannula is inserted through the femoral artery and positioned at the level of the aortic bifurcation. It has a continuous flow centrifugal pump with a maximal rota tion speed of 7,500 revolutions per minute, which can deliver up to 4 l/min. The haemodynamic effects of the TandemHeart are an increase of CO and mean arterial blood pressure (MAP) and a decrease of PCWP, central venous pressure (CVP) and pulmonary artery pressure (PAP). This results in reduced filling pressures in the left and right ventricle, a reduced cardiac workload and a lower oxygen demand.9–11 However, it should be noted, that without direct LV unloading, the increased MAP translates to increased LV afterload, which partially offsets the potential benefit of the lower cardiac workload. The main concerns are the complications (bleeding and limb ischaemia) and the complex insertion procedure. Percutaneous Extracorporeal Membrane Oxygenation ECMO can be achieved percutaneously and is a modified heart–lung machine, which can be used for several days (see Figure 1D). The ECMO system generally consists of a centrifugal pump, a heater and an oxygenator. Venous blood flows from the right atrium into a centrifugal pump and oxygenator and is guided via an outflow cannula in the femoral artery into the descending aorta. The advantage of ECMO over the other percutaneous devices is its ability to provide support in case of RV (RV) failure, to provide higher blood flow rates (up to 4.5 l/min depending on the cannula size) and to oxygenate the blood. Complications associated with ECMO are a systemic inflammatory response, renal failure, limb ischaemia and bleeding complications. Although ECMO can provide substantial haemodynamic support, it also increases both afterload and preload of the left ventricle, increasing the oxygen demand and impeding myocardial protection.12 However, patient transportation with ECMO is relatively easy, which makes it possible to start ECMO support outside the hospital.13
tion speed of 7,500 revolutions per minute, which can deliver up to 4 l/min. The haemodynamic effects of the TandemHeart are an increase of CO and mean arterial blood pressure (MAP) and a decrease of PCWP, central venous pressure (CVP) and pulmonary artery pressure (PAP). This results in reduced filling pressures in the left and right ventricle, a reduced cardiac workload and a lower oxygen demand.9–11 However, it should be noted, that without direct LV unloading, the increased MAP translates to increased LV afterload, which partially offsets the potential benefit of the lower cardiac workload. The main concerns are the complications (bleeding and limb ischaemia) and the complex insertion procedure. Percutaneous Extracorporeal Membrane Oxygenation ECMO can be achieved percutaneously and is a modified heart–lung machine, which can be used for several days (see Figure 1D). The ECMO system generally consists of a centrifugal pump, a heater and an oxygenator. Venous blood flows from the right atrium into a centrifugal pump and oxygenator and is guided via an outflow cannula in the femoral artery into the descending aorta. The advantage of ECMO over the other percutaneous devices is its ability to provide support in case of RV (RV) failure, to provide higher blood flow rates (up to 4.5 l/min depending on the cannula size) and to oxygenate the blood. Complications associated with ECMO are a systemic inflammatory response, renal failure, limb ischaemia and bleeding complications. Although ECMO can provide substantial haemodynamic support, it also increases both afterload and preload of the left ventricle, increasing the oxygen demand and impeding myocardial protection.12 However, patient transportation with ECMO is relatively easy, which makes it possible to start ECMO support outside the hospital.13
Right Ventricular Assist Devices
RV dysfunction has been shown to be a predictor of mortality in STEMI patients with and without CS.14,15 Although the majority of diagnostic and therapeutic approaches for CS are predominantly directed at the left ventricle, there are also assist devices that support the right ventricle. First of all the IABP has been thought to alleviate the RV but clear evidence is absent and due to its limited LV support it is unlikely that the IABP will assist the RV in a meaningful manner. The TandemHeart has an alternative set-up in which it can serve as a RV assist device.16 ECMO can also be used for RV or biventricular failure, as it displaces blood volume from the venous to the arterial circulation and oxygenates the blood. The disadvantage is that LV afterload is increased because there is no direct unloading of the left ventricle. The Impella RP, a specific Impella to support the right ventricle, has recently received a CE-mark in Europe. The Impella RP is placed percutaneously through the femoral vein and advanced in an antegrade fashion across the pulmonic valve into the pulmonary artery (see Figure 2). The Impella RP can provide flow up to 5 l/min for an anticipated duration of 14 days.17