Prognostic Impact
The identification of ischaemia in patients with coronary artery disease (CAD) is likely a key determinant of prognosis.19 Furthermore, reducing the overall ischaemic myocardial burden (i.e. effective re-vascularisation) is effective at long-term improvment prognosis.13,20,21 Contemporary data looking specifically at non-invasive measures of ischaemia suggest that in symptomatic patients with demonstrable ischaemia of greater than 10 % of viable myocardium, a reduction in ischaemic burden by more than 5 % is associated with a significant improvement in overall mortality, as well as a reduction in the incidence of MI.22 This effect is marked most amongst patients who have the greatest ischaemic burden pre-revascularisation.22 It could be inferred, that in patients with at least one CTO and demonstrable ischaemia on a non-invasive test, revascularisation may be of significant clinical benefit.
In patients with a CTO, an ischaemia burden of more than 12.5 % on myocardial perfusion scanning was associated with a greater probability of a successful reduction in ischaemia (greater than 5 % improvement noted in 34.7 % with mild ischaemia vs 86.7 % in severe ischaemia P<0.001) following successful CTO intervention with a sensitivity and specificity of 80 % respectively. Paradoxically, amongst this group of 301 patients, those with a low ischaemia burden before CTO intervention (less than 6.25 %), were more likely to have a increase in ischaemia post PCI.23 This maybe due to restenosis, loss of collateral circulation, loss of side branches, or progression of coronary artery disease in other territories.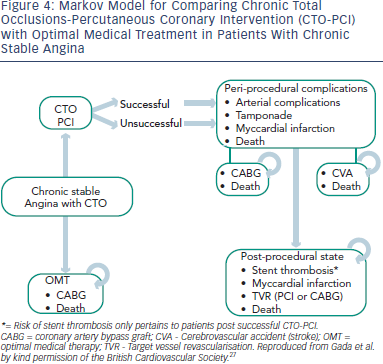
Further evidence that the presence and extent of ischaemia is a determining factor in defining patient prognosis can be derived from examining outcomes after an attempt at PCI. Overall, incomplete revascularisation is associated with worse clinical outcomes when compared to patients in whom complete revascularisation has been achieved.21 The presence of a CTO is a predictor of incomplete revascularisation in those patients treated with PCI. For example, in a cohort of over 11,000 PCI patients treated in the DES era, 69 % (n=7795) were incompletely revascularised. These individuals had a higher mortality rate at 18 months (adjusted HR 1.23, 95 % CI 1.04–1.45), that increased if greater than two vessels (including a CTO) remained unrevascularised (HR 1.44 95 % CI 1.14–1.82, adjusted survival 94.9 % vs 92.9 % p=0.002). Hence, within the context of symptomatic angina, successful CTO intervention which leads to complete revascularisation (and relief of underlying ischaemia) may offer a substantial mortality benefit.20 It is also important to appreciate that CABG is not certain to produce complete revascularisation. For example, in the Syntax trial CTO subset, successful bypass grafting to an occluded arterial segment occurred in only 69 %, with complete revascularisation (in the context of a CTO) being achieved in only 49 %24, albeit by a very strict definition. Interpretation of these data requires care, of course.
There remains a paucity of a large scale randomised trial data that specifically addresses the outcomes after successful ischaemia-driven CTO intervention. The bulk of data currently available to support CTO interventional practice is derived from registries that compare patients in whom CTO intervention has been successful with those in whom it has not. For example, when comparing 582 successful CTO procedures with 282 cases in which revascularisation was not achieved, Jones et al. report a 12.7 % decrease in mortality at five years.25 A metaanalysis of data available up until 2010, including over 7,000 patients across 13 studies found a 3.2 % lower mortality with successful CTO intervention13 (14.3 % vs 17.5 % OR 056, 95 % CI 0.43–0.72). In addition, a significantly lower rate of CABG was observed (OR 0.22, 95 % CI 0.17–0.27), but no differences in MI or other major adverse cardiac events were noted. Overall, successful CTO intervention does appear to offer a prognostic advantage, but only when compared to those in whom a CTO procedure was unsuccessful. These data provide circumstantial evidence at best: a randomised trial of CTO PCI versus optimal medical therapy (OMT) with an intention-to-treat analysis is required for definitive proof of benefit.
The reasons behind the difference between successful and unsuccessful CTO PCI remain undefined. Certainly, the differences observed can only partly be explained by adverse events in the unsuccessful PCI arms, the rates of which are broadly similar across reported data.13 The observed prognostic benefit may relate partly to a reduction in ischaemia driven arrhythmia,18 and possibly improved outcome amongst patients who go on to have an acute myocardial infarction of a non-CTO artery, on the basis of the “double jeopardy” theory. This was demonstrated in one large series of over 3,200 STEMI patients where the presence of a CTO, distinct from the culprit artery, was an independent predictor of 30-day mortality (HR 3.6, 95 % CI 2.6– 4.7; p<0.01). In those alive at 30 days, the presence of a CTO remains a predictor of overall five year mortality.26
To summarise, although the current data are encouraging, Despite these favourable indicators, CTO interventional practice is still lacking a large scale randomised trial to formally demonstrate prognostic benefit for the procedure. However, to answer this criticism, two large scale randomised trials comparing CTO intervention with optimal medical therapy are ongoing, EURO-CTO and DECISIONCTO, both of whom are still recruiting and due to report in 2017 & 2018 respectively.