Diabetes Mellitus and Heart Failure – A Bidirectional Relationship
From Diabetes Mellitus to Heart Failure
The concept that DM can cause or precipitate HF has been generated even from the Framingham study 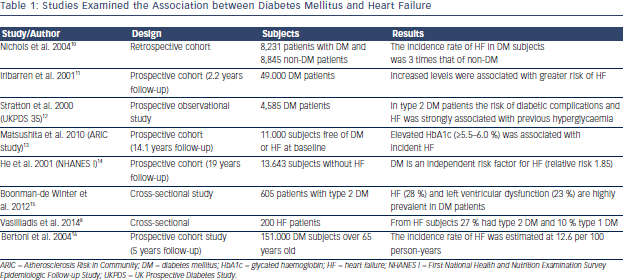 who estimated that men and women with DM have a two and four times, respectively, increased risk to develop HF compared with non-DM subjects.9 Epidemiological studies have confirmed this relationship and revealed that impaired glucose tolerance, increased serum glucose levels and glycated haemoglobin levels are associated not only with incidence of systolic HF but also with the prevalence of diastolic dysfunction (see Table 1). Accordingly, guidelines have accepted DM and metabolic syndrome as risk factors for HF,17 and the term diabetic cardiomyopathy was used to define either systolic or diastolic left ventricular dysfunction in otherwise healthy diabetic persons in the absence of clinically significant coronary, valvular or hypertensive disease.18 Thus, patients with DM can be categorised in the stage A of the American College of Cardiology Foundation (ACCF)/American Heart Association (AHA) classification of HF, meaning that diabetic subjects are at high-risk for HF but without structural heart disease or symptoms of HF. Therapeutic interventions aim to modify risk factors and guidelines draw attention to monitor and treat DM as early as possible.17
who estimated that men and women with DM have a two and four times, respectively, increased risk to develop HF compared with non-DM subjects.9 Epidemiological studies have confirmed this relationship and revealed that impaired glucose tolerance, increased serum glucose levels and glycated haemoglobin levels are associated not only with incidence of systolic HF but also with the prevalence of diastolic dysfunction (see Table 1). Accordingly, guidelines have accepted DM and metabolic syndrome as risk factors for HF,17 and the term diabetic cardiomyopathy was used to define either systolic or diastolic left ventricular dysfunction in otherwise healthy diabetic persons in the absence of clinically significant coronary, valvular or hypertensive disease.18 Thus, patients with DM can be categorised in the stage A of the American College of Cardiology Foundation (ACCF)/American Heart Association (AHA) classification of HF, meaning that diabetic subjects are at high-risk for HF but without structural heart disease or symptoms of HF. Therapeutic interventions aim to modify risk factors and guidelines draw attention to monitor and treat DM as early as possible.17
Pathophysiological Connections
Despite the close relationship of these two conditions the difficulties in making a causal pathophysiological connection between DM 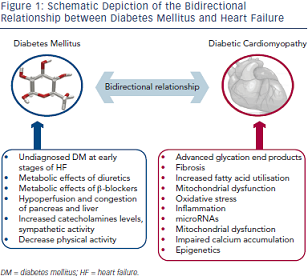 and HF are formidable as we have to distinguish between insulin deficient and insulin resistant DM and between systolic HF, diastolic HF or HF caused by other aetiologies such as conduction disturbances, tachycardiomyopathy, HF of valvular aetiology, etc. (see Figure 1).
and HF are formidable as we have to distinguish between insulin deficient and insulin resistant DM and between systolic HF, diastolic HF or HF caused by other aetiologies such as conduction disturbances, tachycardiomyopathy, HF of valvular aetiology, etc. (see Figure 1).
Advanced glycation end products, which are formed in DM subjects after non-enzymatic reaction between protein and sugar residues, can possibly explain the link between DM and HF.19 Advanced glycation end products are increased in patients with chronic HF, they correlate inversely with left ventricular ejection fraction and are related to the severity and prognosis of the disease.20,21 Advanced glycation end products upregulate the hypertrophy-associated genes in cardiomyocytes via the activation of dendritic cells and may be responsible for the hypertrophic and fibrotic phenotype in DM subjects.22
A plethora of other mechanisms have also been proposed. Diabetes-related alterations in the expressions of some calciumassociated proteins may lead to progressive intracellular decay of calcium and in the development of diabetic cardiomyopathy.23 In rats with diabetic cardiomyopathy there are also decreased serum and myocardial levels of adiponectin implying a possible connection between this anti-inflammatory protein and HF.24 Hearts in humans with DM are characterised by increased fatty acid metabolism and oxidation, which is considered a pathophysiological mechanism in the development of HF.25 Moreover, animal studies have revealed increased myocardial levels of cardiotoxic inflammatory cytokines (tumour necrosis factor-alpha, intereukin-6, etc.) in diabetic models.26
Hyperinsulinaemia also impairs phosphatidylinositol 3-kinases pathway and can precipitate myocardial dysfunction.27 Furthermore, accumulation of reactive oxygen species affects the coronary circulation and causes myocardial hypertrophy and fibrosis.28 Carbonic anhydrases have been shown to play a major role in diabetic microangiopathy. Recently, carbonic anhydrases I and II were found elevated in myocardial tissues from post-infarction HF patients with DM. They can induce cardiomyocyte hypertrophy and death in vitro, which are prevented by sodium-hydrogen exchanger-1 inhibition.29 MicroRNAs are also differentially expressed in myocardial tissues from subjects with diabetic cardiomyopathy compared with non-diabetic HF, and specific patterns have been recognised.30–32 These data may provide new targeted treatment of diabetic HF.
We also notice that diabetes frequently precedes coronary heart disease,33 chronic kidney disease34 and hypertension,35 which are major risk factors and account for the majority of HF cases,5 further explaining the close relationship between DM and HF observed in epidemiological studies.
Clinical and Epidemiological Connections
Further to pathophysiological connections, several clinical data have confirmed the detrimental impact of DM in HF course and prognosis. DM predicts readmissions of HF patients36 and increases mortality in subjects with left ventricular dysfunction.37 Elevated troponin levels in patients with DM are also associated with increased HF and cardiovascular mortality.38 Recently, it was also confirmed that DM can be used in a model to predict chronic HF patients at risk of hospitalisation.39 Moreover, in patients with type 2 DM, glycated haemoglobin significantly predicts future HF hospitalisation independently of baseline b-type natriuretic peptide (BNP) level or echocardiographic parameters.40
From Heart Failure to Diabetes Mellitus
The dual nature of the relationship of HF and DM is supported by prospective cohort studies. DM was developed in 29 % of HF subjects compared with 18 % of matched control subjects during a three-year follow-up study.41 Moreover, in a cohort of 50.874 patients, HF severity (as determined by loop diuretic dosages) predicts the risk of developing diabetes after myocardial infarction.42
Nevertheless, the mechanisms precipitating DM and HF are not well-studied. Firstly, we have to notice the possibility that the increased incidence of DM during the course of HF may be an epiphenomenon of the lenient monitoring for impaired glucose metabolism, with glycated haemoglobin and with oral glucose tolerance tests in the early stages of HF. Accordingly, guidelines emphasise the importance of proper diagnosis of DM in HF subjects.5
We can also hypothesise that the decreased physical activity in HF patients may lead to decreased insulin sensitivity and to compensatory insulin requirements and hyperglycaemia. In addition, increased catecholamines levels and sympathetic activity stimulate gluconeogenesis and glycogenolysis.43 Indeed, there is a decrease in insulin sensitivity according to New York Heart Association (NYHA) functional status of HF patients.44
Furthermore we can hypothesise that the haemodynamic consequences accompanying HF (decreased forward blood flow and increased central venous pressure) lead to hypoperfusion and congestion of the pancreas and liver, which may impair their ability to regulate metabolic homeostasis. Confirmatory data are provided by a recent study, which concludes that left ventricular assist devices improve blood glucose control in DM patients.45
Finally, we have to notice the possible adverse effects of established HF treatments, such as beta (β)-blockers and diuretics in blood glucose control.46