iFR is not Independent of Hyperaemia
iFR assumes that microvascular resistance is already constant and minimal in the wave-free period. Both the VERIFY study and the analysis from Johnson et al. clearly demonstrate that this 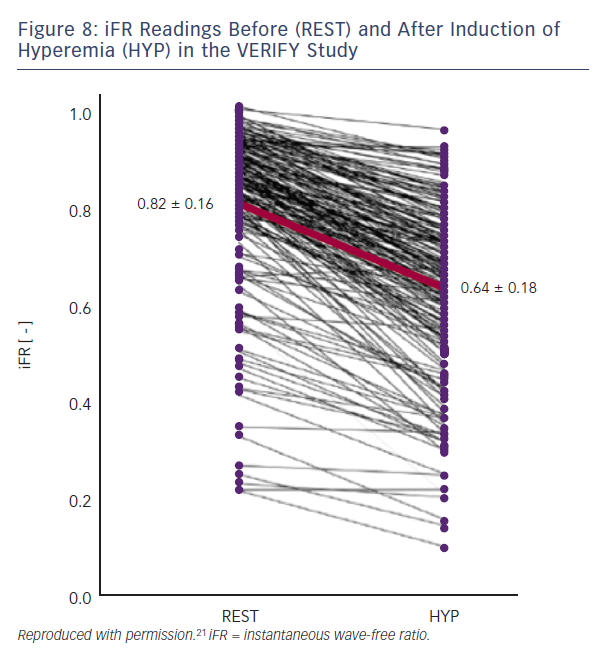
Loss of Diagnostic Accuracy
When binary cut-off values are used for decision making, as in the ongoing randomised trials of iFR guidance versus FFR guidance, DEFINE FLAIR (NCT02053038) and iFR Swedeheart (NCT02166736), the currently available data clearly confirm that avoiding the use of adenosine altogether translates into a different treatment decision in 20 % of lesions compared with an FFR for all strategy. Given wide enough non-inferiority margins and the knowledge that 80 % of the population recruited will have convergent iFR and FFR results, it is likely that these studies will show non-inferiority. However, such a result would conceal a mix of both inappropriate PCI and incomplete revascularisation in the iFR-guided group compared with an FFR-guided strategy.
To the authors’ knowledge, there are no randomised controlled trials comparing the hybrid iFR/FFR decision tree with an FFR for all strategy. SYNTAX II (NCT02015832) does employ this option but is an observational study. The currently available data are less comprehensive than for binary cut-off decision-making but avoiding the use of adenosine in between 50–60% of patients may translate into a different treatment decision in around 10 % of patients. Again, this will conceal a mixture of inappropriate PCI and incomplete revascularisation.
Beyond these concerns, if iFR was employed at the time of initial diagnostic angiography, as in the RIPCORD study, then patients will likely have divergent management decisions compared with an FFR for all strategy.14 This will translate not just into different patterns of stent implantation but different overall management in terms of which patients undergo revascularisation and by which technique.
Whether employed in patients already scheduled for PCI or within a binary cut-off or hybrid strategy there is another major concern about using iFR or indeed any resting index of stenosis severity to determine the need for revascularisation. The flow increase associated with hyperaemia when interrogating a large-calibre vessel with a proximal stenosis is much greater than that of a small diameter vessel supplying a small myocardial territory.35 This means that some prognostically important lesions may exhibit small resting pressure gradients and therefore non-ischaemic iFR results which become manifestly ischaemic FFR results during hyperaemia. On the basis that some of these lesions have ‘non-ischaemic’ CFR values >2.0, it has been argued that the FFR result is false and the iFR result is correct. Thus, in the study by van de Hoef et al, half of the patients with discordant iFR and FFR values were ‘ischaemic’ according to the combined reference standard of MPS/HSR, and the other half were ‘non-ischaemic’.20 Notwithstanding the limitations of this study as described above, this indicates that approximately 50 % of treatment decisions will likely be incorrect. Furthermore, a CFR >2.0 does not in any way exclude myocardial ischaemia and may still be too low for a given coronary vessel and myocardial territory at high myocardial oxygen demand.